Thyroid (USP) is an Active Pharmaceutical Ingredient (API) used to treat thyroid dysfunction. It is also known as Desiccated Thyroid extract or Natural Desiccated Thyroid. It is the active ingredient of medications such as Armour Thyroid or NP Thyroid. Thyroid (USP) is made of pigs’ thyroid glands.
Thyroid dysfunction is a condition that can affect your body’s ability to produce enough thyroid hormones. Thyroid (USP) is used to treat a number of conditions, including:
- hypothyroidism
- primary hypothyroidism
- Graves’ disease
- Hashimoto’s thyroiditis
- fibromyalgia
Since the 1980s’ The United States Pharmacopeia (USP) has defined what Thyroid used in pharmaceutical products is. This means that the quality and purity of thyroid hormones are important and that they must meet specific safety and efficacy standards.

Table of Contents
ToggleHow many thyroid hormones does Thyroid (USP) have?
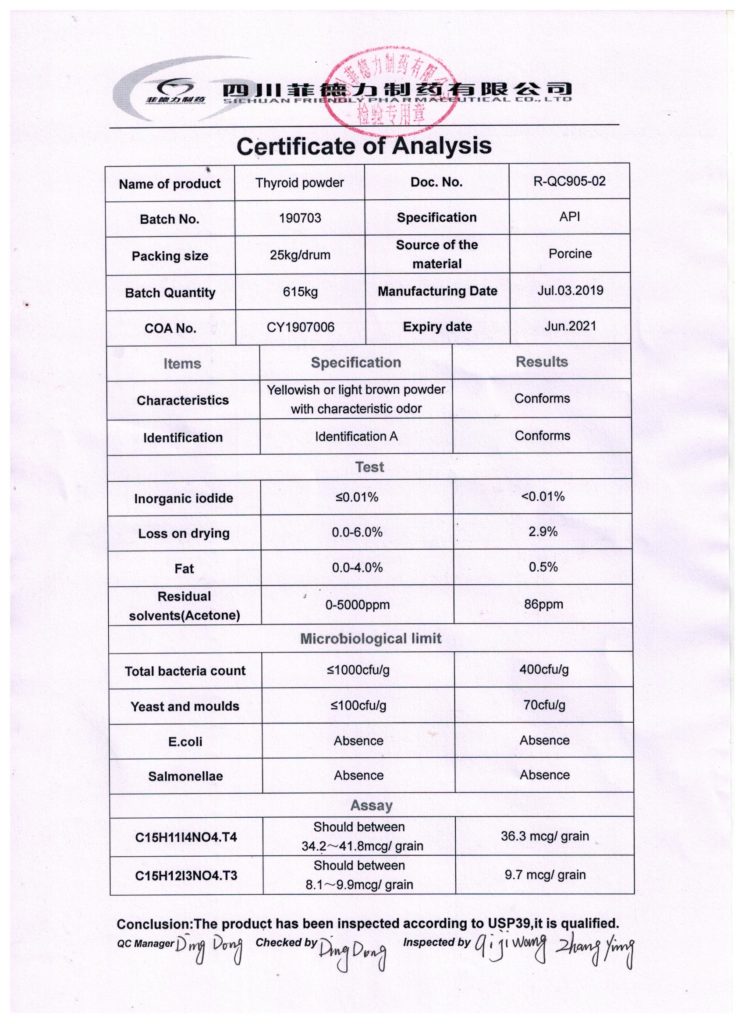
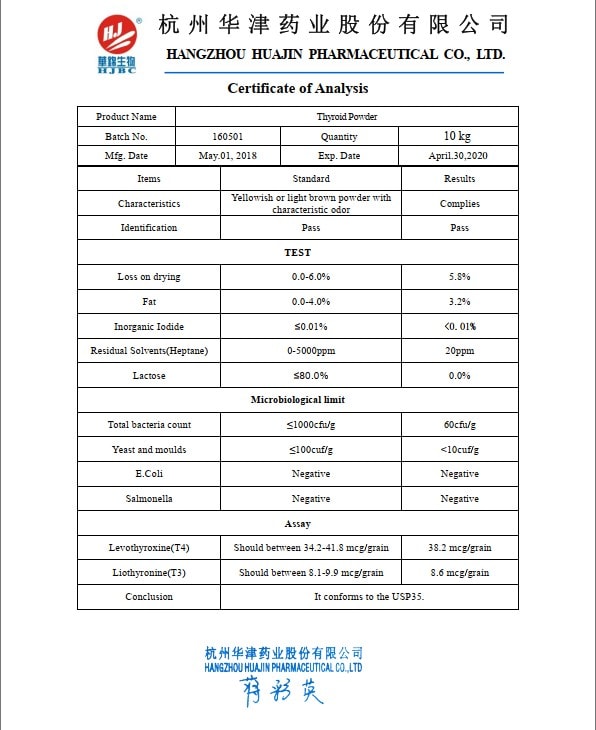
The “ideal” Thyroid (USP) contains 38 mcg of T4 and 9 mcg of T3 per grain (60 or 65 mg of Thyroid powder). However, due to the natural origin of the source, 10% difference in hormones levels is allowed.
That means that Thyroid USP potentially could contain as little as 35.2 mcg of T4 and as little as 8.1 mcg of T3. On the higher end, it can contain up to 41.8 mcg of T4 and 9.9 mcg of T3. The range is:
- 34.2-41.8 mcg of T4 per grain (60 or 65 mg of Thyroid powder)
- 8.1-9.9 mcg of T3 per grain (60 or 65 mg of Thyroid powder)
Where is Thyroid USP used?
Thyroid USP powder is used in many medicines such as:
- Armour Thyroid
- NP Thyroid
- WP Thyroid
- Nature-Throid
- ERFA Thyroid
- Compounded pharmacies’ thyroid
- generic Thyroid
It is used in a selected few, quality thyroid supplements:
- Real Thyroid
- Vitalithy
Is Thyroid USP brand name?
There are many different brands of natural thyroid medications on the market, each with its own specific name. However, the active ingredient in all of these medications is the same: Thyroid USP. The thyroid USP is a name given to an API (Active Pharmaceutical Ingredient) used in many brands of natural thyroid medications such as Armour Thyroid or NP Thyroid. This API is derived from pigs’ thyroid gland itself and is considered to be the most effective form of thyroid medication available.
There are a few reasons why the thyroid USP is considered to be the best option. First, it is derived from the thyroid gland itself, which means that it is more effective than other forms of synthetic thyroid medication. Additionally, Thyroid USP is more stable than other forms of thyroid medication and is less likely to cause side effects. Finally, the Thyroid USP is easier to take than other forms of thyroid medication, which makes it more convenient for patients.
http://www.pharmacopeia.cn/v29240/usp29nf24s0_m83400.html
Thyroid USP Monography:
Packaging and storage—
Preserve in tight containers.
USP Reference standards ![]() 11
11![]() —
—
USP Levothyroxine RS. USP Liothyronine RS.
Identification—
The retention times of the peaks for liothyronine and levothyroxine in the chromatogram of the Assay preparation correspond to those in the chromatogram of the Standard preparation, as obtained in the Assay.
Microbial limits ![]() 61
61![]() —
—
It meets the requirements of the tests for absence of Salmonella species and Escherichia coli.
Loss on drying ![]() 731
731![]() —
—
Dry it in vacuum at 60![]() for 4 hours: it loses not more than 6.0% of its weight.
for 4 hours: it loses not more than 6.0% of its weight.
Limit of inorganic iodides—
Extracting solution—
Prepare a 1 in 100 solution of sulfuric acid in water.
Reference solution—
Dissolve an accurately weighed quantity of potassium iodide in water to obtain a stock solution containing 0.131 mg, equivalent to 0.100 mg of iodide, per mL. Transfer 1.0 mL of this stock solution into a 100-mL volumetric flask, dilute with Extracting solution to volume and mix. Each mL of the Reference solution contains 1.0 µg of iodide. [NOTE—Prepare this solution on the day of use.]
Test solution—
Transfer 1.00 g, or proportionately less if the iodine content is greater than 0.2%, of Thyroid to a beaker, add 100.0 mL of Extracting solution, and sonicate for 5 minutes.
Electrode system—
Use an iodide-specific, ion-indicating electrode and a silver-silver chloride reference electrode connected to a pH meter capable of measuring potentials with a minimum reproducibility of ±1 mV (see pH ![]() 791
791![]() ).
).
Procedure—
Transfer the Reference solution to a beaker containing a magnetic stirring bar. Rinse and dry the electrodes, insert in the solution, stir for 5 minutes or until the reading stabilizes, and read the potential, in mV. Repeat this process using the Test solution. The requirements of the test are met if the Test solution has a higher potential, in mV, than the Reference solution: the limit is 0.01%.
Residual solvents ![]() 467
467![]() :
:
meets the requirements.
Assay—
Mobile phase—
Prepare a degassed and filtered mixture of water, acetonitrile, and phosphoric acid (650:350:5). Make adjustments if necessary (see System Suitability under Chromatography ![]() 621
621![]() ).
).
Reducing buffer solution—
Freshly prepare a solution in 0.11 M sodium chloride that is 0.04 M with respect to tris(hydroxymethyl)aminomethane and 0.05 M with respect to methimazole. Adjust, if necessary, with 6 N hydrochloric acid or 0.1 N sodium hydroxide to a pH of 8.4 ± 0.05.
Proteolytic enzyme—
Freshly prepare a solution containing 15 mg of bacterial protease* in each 5 mL of Reducing buffer solution.
Enzyme deactivating solution—
Prepare a 1 in 100 mixture of phosphoric acid in acetonitrile.
Standard preparation—
[NOTE—Protect solutions from light.] Transfer accurately weighed quantities of about 9 mg of USP Liothyronine RS and about 38 mg of USP Levothyroxine RS to a 100-mL volumetric flask, add 50 mL of a mixture of water, acetonitrile, and ammonium hydroxide (500:500:1), and swirl to dissolve. Dilute with a mixture of water and acetonitrile (1:1) to volume, and mix (stock solution). On the day of use, pipet 5 mL of the freshly prepared stock solution into a 250-mL volumetric flask, dilute with Reducing buffer solution to volume and mix to obtain a solution having known concentrations of about 1.8 µg of liothyronine per mL and about 7.6 µg of levothyroxine per mL. Pipet 5 mL of this solution into a screw-capped 16- × 125-mm culture tube. Pipet 2 mL of Enzyme deactivating solution into the tube, place the cap on the tube, and shake the mixture vigorously.
Assay preparation—
Transfer an accurately weighed portion of finely powdered Thyroid, equivalent to about 38 µg of levothyroxine, to a screw-capped 16- × 125-mm culture tube that previously has been flushed with nitrogen. Taking precautions to avoid unnecessary exposure to air, pipet 5 mL of Proteolytic enzyme into the tube. Allow nitrogen to flow gently over the mixture for 5 minutes. Place the cap on the tube, mix to disperse the contents, and place in a covered water bath maintained at a temperature of 37 ± 1![]() for 28 hours. Protect the contents of the tubes from light. Examine occasionally, and mix as necessary to ensure dispersion. At the end of the incubation period, pipet 2 mL of Enzyme deactivating solution into the tube, place the cap on the tube, mix vigorously, and centrifuge at about 2000 rpm for 5 minutes. Filter the supernatant through a 0.45-µm porosity filter, discarding the first 1 mL of the filtrate.
for 28 hours. Protect the contents of the tubes from light. Examine occasionally, and mix as necessary to ensure dispersion. At the end of the incubation period, pipet 2 mL of Enzyme deactivating solution into the tube, place the cap on the tube, mix vigorously, and centrifuge at about 2000 rpm for 5 minutes. Filter the supernatant through a 0.45-µm porosity filter, discarding the first 1 mL of the filtrate.
Chromatographic system (see Chromatography ![]() 621
621![]() )—
)—
The liquid chromatograph is equipped with a 230-nm detector and a 4.6- × 25-cm column that contains packing L1. The flow rate is about 1.5 mL per minute. Chromatograph the Standard preparation, and record the peak responses as directed for Procedure: the tailing factors for the liothyronine and levothyroxine peaks are not more than 1.8, and the relative standard deviation for replicate injections is not more than 2.0%.
Procedure—
Separately inject equal volumes (about 200 µL) of the Assay preparation, and the Standard preparation, record the chromatograms and measure the responses for the major peaks. Calculate the quantity, in µg, of liothyronine (C15H12I3NO4) and levothyroxine (C15H11I4NO4) in the portion of Thyroid taken by the formula:
in which C is the concentration, in µg per mL, of the corresponding USP Reference Standard in the Standard preparation, and rU and rS are the peak responses for the corresponding analytes obtained from the Assay preparation and the Standard preparation, respectively.
*A suitable grade is available as “Pronase” (Catalog number 53702) from Calbiochem-Behring, P. O. Box 12087, San Diego, CA![]() 92112.
92112.
Auxiliary Information—
Staff Liaison : Larry N. Callahan, Ph.D., Scientist
Pharmacopeial Forum : Volume No. 28(3) Page 789
4 thoughts on “What is Thyroid (USP)?”
Pingback: Can you Buy NP Thyroid online without prescription? - Thyroid And Metabolism
Pingback: Nature-Throid ingredients - Thyroid And Metabolism
Pingback: What is the difference between Real Thyroid and Thiroyd, Thyroid-S or TR Thyroid? What are the similarities? - Thyroid And Metabolism
Pingback: Armour Thyroid: Weight gain as a side effect? - Thyroid And Metabolism
Comments are closed.